 In that case, the nuclear particles will be more tightly bound than they were in the lighter nuclei, and that decrease in mass comes off in the form of energy, according to the Albert Einstein relationship. Suppose the combined nuclear mass is less than that of iron at the peak of the binding energy curve. If light nuclei are forced together, they will fuse with a yield of energy because the mass of the combination will be less than the sum of the masses of the individual nuclei. In nuclear physics, nuclear fusion is a nuclear reaction in which two or more atomic nuclei collide at very high energy and fuse together into a new nucleus, e.g., helium. But not all the total energy can be recovered in a reactor. The total energy released in fission can be calculated from binding energies of the initial target nucleus to be fissioned and binding energies of fission products. In reactor physics, neutron-induced nuclear fission is the process of the greatest practical importance. The decay process is called spontaneous fission, and it is a very rare process. Nuclear fission is either a nuclear reaction or a radioactive decay process in nuclear physics. The fission process often produces free neutrons and photons (in the form of gamma rays) and releases a large amount of energy. 
Nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts (lighter nuclei). Whether the reaction is exothermic or not depends on the binding energy of the resulting nuclei. Nuclear fusion is a nuclear reaction in which two or more atomic nuclei collide at very high energy and fuse together into a new nucleus.Nuclear fission is a nuclear reaction in which the nucleus of an atom splits into smaller parts (lighter nuclei).  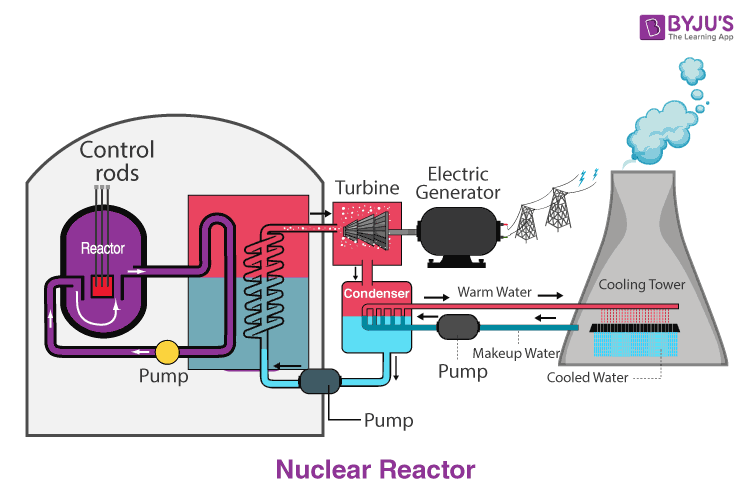
Nuclear fission and nuclear fusion are different types of reactions that release energy (when exothermic) due to the formation of nuclei with higher nuclear binding energy. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
